Trust Us To Lead The Way In Certification And Compliance
Knowledge And Expertise
Thorough Understanding Of The Framework, Its Requirements, And Best Practices For Implementation
Proven Track Record
Successful Track Record Of Helping Clients Achieve Compliance, With Positive Client Testimonials And Case Studies.
Strong Project Management Skills
Ensure The Compliance Engagement Runs Smoothly And Is Completed On Time And Within Budget.
Experienced Team
Possession Of Experienced Professionals, Including Auditors, Consultants, And Technical Experts
Exceptional Customer Service
Committed To Excellent Customer Service With Clear Communication, Responsive Support, And A Focus On Satisfaction.
Competitive Pricing
We Prioritize Delivering High-Quality Services With Competitive Pricing That Provides Exceptional Value To Our Clients
FAQs
FREQUENTLY ASKED
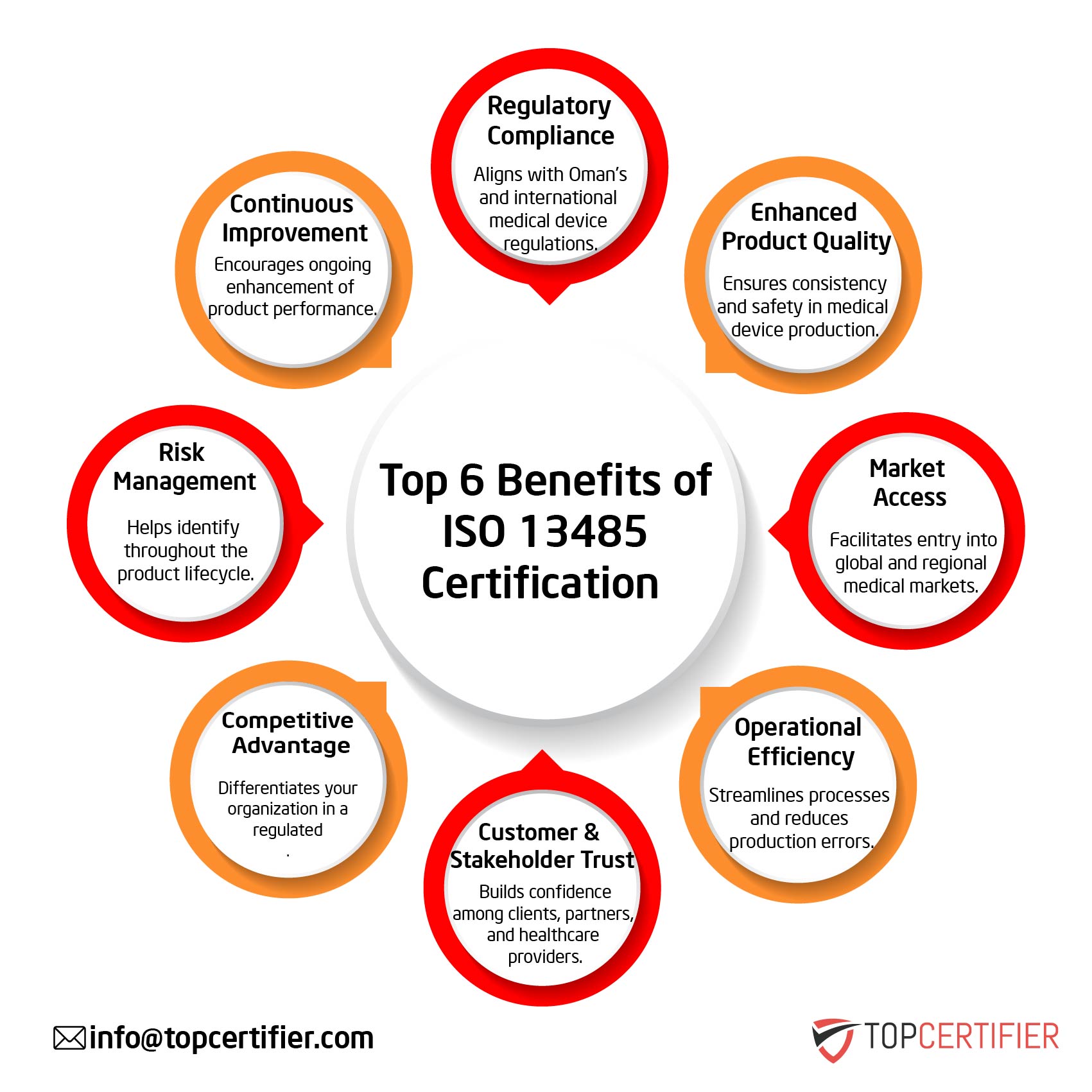
ISO 13485 certification is an internationally recognized Quality Management System (QMS) standard specifically designed for the medical device industry. It helps organizations ensure product safety, regulatory compliance, and consistent quality throughout the medical device lifecycle.
Any organization involved in the design, manufacture, distribution, installation, or servicing of medical devices in Bangladesh can apply for ISO 13485 certification, including manufacturers, suppliers, distributors, and service providers.
ISO 13485 certification is not legally mandatory, but it is highly recommended for organizations seeking regulatory approval, market access, and compliance with national and international medical device regulations.
ISO 13485 certification improves product quality, ensures patient safety, enhances regulatory compliance, reduces risks, and increases customer confidence in medical devices and related services.
ISO 13485 is widely used by medical device manufacturers, healthcare equipment suppliers, diagnostic laboratories, pharmaceutical support companies, and medical device distributors.
The ISO 13485 certification process usually takes 4 to 10 weeks, depending on organization size, scope, readiness level, and regulatory requirements.
The cost of ISO 13485 certification in Bangladesh depends on company size, number of products, compliance scope, and audit complexity. Contact us for a customized quotation.
Yes, ISO 13485 supports compliance with medical device regulations by establishing documented processes, risk management, and quality controls aligned with regulatory expectations.
Professional ISO consultants assist with gap analysis, QMS documentation, risk management, internal audits, and certification audits—ensuring a smooth and compliant ISO 13485 certification process.